 
The cycling of water in and out of the atmosphere is a significant aspect of the weather patterns on Earth. Precipitation hardening, also called age hardening or particle hardening, is a heat treatment technique used to increase the yield strength of malleable materials, including most structural alloys of aluminium, magnesium, nickel, titanium, and some steels, stainless steels, and duplex stainless steel. The water falling on land collects in rivers and lakes, soil, and porous layers of rock, and much of it flows back into the oceans, where it will once more evaporate. The water cycle describes how water evaporates from the surface of the earth, rises into the atmosphere, cools and condenses into rain or snow in clouds, and falls again to the surface as precipitation. In precipitation, the solute particles find each other and form a solid together. In dissolution, the solute particles separate from each other and are surrounded by solvent molecules. It is the opposite of dissolution or solvation. Frequent and detailed measurements help scientists make models of and determine changes in Earth’s water cycle. Precipitation is the process of a compound coming out of solution. Knowing where it rains, how much it rains and the character of the falling rain, snow or hail allows scientists to better understand precipitation’s impact on streams, rivers, surface runoff and groundwater. 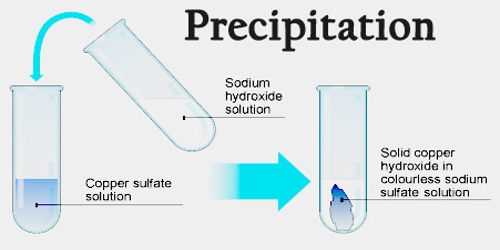
Precipitation reactions are also useful to extract elements, like magnesium, from seawater.Precipitation is a vital component of how water moves through Earth’s water cycle, connecting the ocean, land, and atmosphere. The insoluble product is called a precipitate and can be seen. For example, a barium nitrate solution will react with sulfate ions to form a solid barium sulfate precipitate, indicating that it is expected that sulfate ions are present. A precipitation reaction is a reaction that yields an insoluble producta precipitatewhen two solutions are mixed. A precipitation reaction is one in which substances in solution are mixed and an insoluble product is made. Precipitate development is valuable in the detection of the type of cation in a salt. Subsequently, the precipitate may easily be separated by filtration/decanting or centrifugation. Antisolvent is added - This radically drops the solubility of the desired product. the amount of rain, snow, hail, etc., that has fallen at a given place within a given period, usually expressed in inches or centimeters of water.precipitate To cause to fall as a sediment to the bottom of a vessel reduce from a state of solution to a solid form, as by means of a reagent or chemical force. Using a supersaturated solution - Without sufficient force of gravity (settling) to bring the solid particles together, the precipitate remains in suspension. precipitate To cast down headlong fling from a precipice or height hurl downward.The chemical that leads the solid to form is called the precipitant. For example, a solution containing Ba 2+ and SO42- ions may form a. Temperature of a solution is lowered - The lower temperature reduces the solubility of a salt, resulting in its precipitation as a solid. A precipitate may form when solutions are mixed which contain the appropriate ions.Two soluble salts react in solution to form one or more insoluble products.Powders derived from precipitation are known as flowers. The precipitate-free liquid remaining above the solid is called the supernate or supernatant. Precipitate is referred to as a pellet after sedimentation when using a centrifuge to press it into a compact mass. When the reaction occurs in a liquid solution, the solid formed is called the precipitate. Precipitation is the creation of a solid in a solution or inside another solid during a chemical reaction or by diffusion in a solid. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
